A review of some of the most relevant elements of the 2021 surviving sepsis campaign for the emergency physician.
Definitions
Our operative definition of sepsis & septic shock are the sepsis-3 definitions [1].
Sepsis: “Sepsis is a life threatening organ dysfunction caused by a deregulated host response to infection.”
Septic Shock: Diagnosis is based on three criteria
- Diagnosis of sepsis
- Vasopressors required to maintain MAP ≥ 65
- Lactate ≥ 2 mmol/L after fluid resuscitation
Diagnosis
The diagnosis of sepsis is thought to hinge on the presence of an infection and at least 2 qSOFA Criteria which includes:
- Altered mental status (GCS<15)
- Tachypnea (RR>22)
- Hypotension (SBP<100)
It is worth noting that there are some issues with qSOFA, and so it’s use for the diagnosis of sepsis is likely transitory while we wait for Sepsis-4. Of note, qSOFA was derived from SOFA which is an ICU-based score used for predicting mortality. Similarly to SOFA, qSOFA was also found to predict mortality; however, wasn’t analyzed regarding its effectiveness as a screening tool. Three major meta-analyses have since looked at the utility of qSOFA as a screening tool and all proposed a strong recommendation against using qSOFA as a single screening tool for sepsis or septic shock [2–4]. As such, it is recommended to use a tandem approach where for a SIRS+ patients, you must entertain sepsis as a diagnosis and apply the qSOFA score to them. Should they score positive on qSOFA, then the patient is at higher risk of poor outcomes, and likely warrants closer attention.
Sepsis Care Bundles
A major point of contention surrounding the 2018 surviving sepsis campaign update related to the 1 hour sepsis bundle [5].
The bundle consisted of:
- Measure a lactate
- Obtain blood cultures
- Administer broad spectrum antibiotics
- Rapid administration of 30cc/kg crystalloid for hypotension or lactate >4
- Apply vasopressors MAP >= 65
This bundle had a target of 1 hour to administration, with the clock starting at the time of triage in the ED.
Issues
The bundle did not discriminate between the presence or absence of shock. This was problematic, as this 1 hour benchmark was unlikely achievable in those with less severe / classic presentations of sepsis, such as the elderly. Moreover, in an era of crowding and hallway medicine, concerns were raised that funding metrics may now be tied to this unrealistic benchmark. There were fears of aggressive presumptive treatment of patients with any features possibly consistent with sepsis at the door of the ED “for the sake of funding”. Finally, this change seemed like a set-up for early diagnostic closure and it’s associated harms.
To really drive home how potentially problematic the 1 hour bundle was, I want you to envision the following patient, Gladys.
|
When seeing this patient, what would your approach be to fluid resuscitation?
Your answer likely involves a clinical assessment and contains nuance based on the provision of more data. That’s wonderful, and exactly what our patient’s need. I want to highlight that Gladys is a patient with new decompensated heart failure. With an appropriate clinical evaluation she would likely do very well, the reason for her symptoms would be identified and she would be appropriately managed. I want you all to pause for a moment to consider how Gladys would do when empirically given 2.5L of fluid to meet a 1 hour bundle target. The 1 hour bundle embraced the protocolized, data driven essence of EGDT while stepping away from invasive monitoring that has since been shown to be without benefit. No longer is there an emphasis on aggressive transfusion targets, central venous pressures or central venous oxygen sats – this is objectively a step in the right direction and an improvement over prior recommendations.Click here for further discussion…
While the overzealous recommendations of 2018 may have been a bit of a misstep, they also presented an opportunity for iterative improvement in the form of the 2021 guideline. The 2021 surviving sepsis campaign has redefined the timelines revolving around their bundle therapy based on feedback they received. The new sepsis bundles have an emphasis on the clinical context of a patient’s presentation. A huge step in the right direction.
The New 2021 Surviving Sepsis Campaign Time Targets
When trying to determine the timeline over which you should be treating your septic patient’s, there are essentially 3 questions you must answer:
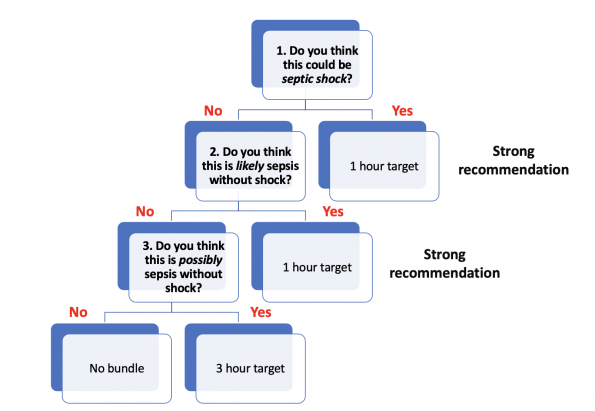
It is important here to keep in mind that the second question isn’t asking if this patient has an infection, it is asking if this person likely has a dysregulated host response to infection. While some of that is guided by bloodwork and signs of organ injury, those investigations take time. This is your clinical gestalt on who warrants an expedited treatment pathway. In my mind, these are mostly the hemodynamically stable patients that are SIRS + and qSOFA + or clinically appear worse than their vital signs would suggest.
For those where sepsis is still a potential diagnosis (pre-test probability low-moderate), a 3 hour window is provided for rapid investigation, evaluation and observation. If investigations increase your suspicion of sepsis in this patient, or if the clinical situation changes, administer bundle within 3 hours. If following this evaluation you are reassured that the patient is not septic, proceed as you would normally [6].
Now that we’ve decided who gets a bundle and what timeline they receive it over, let’s discuss the nuances of the current sepsis bundle.
The New 2021 Surviving Sepsis Campaign Care Bundle
The following are components are each considered in the new 2021 Surviving Sepsis Campaign:
- Measure Lactate
- Obtain Microbial Cultures
- Administer antimicrobials
- Administer Crystalloids
- Initiate vasopressor therapy as needed
_________________________
1. Measure Lactate
Lactate measurement is an important element of sepsis care. The current surviving sepsis campaign highlights three principal roles for lactate:
Diagnosis, Prognosis and Evaluation of Resuscitation.
Diagnosis
As a reminder – lactate is now an integral part of the third universal definition of septic shock.
Prognosis
There is a well established association between elevated lactate measurements and mortality across numerous studies.
While this only represents one data point in anticipating a patient’s clinical course, it’s a useful one. Particularly given that by drawing a lactate level early, we acquire a picture of the patient’s physiology pre-treatment to direct both our treatment and that of our ICU colleagues.
Resuscitation Evaluation
The 2021 surviving sepsis campaign recommends guiding resuscitation to decrease lactate when it is elevated at presentation.
Additionally, there is also a provided recommendation to use capillary refill time to guide resuscitation as an adjunct to other measures of perfusion, such as clinical exam and lactate.
For more details check out this blog post, or this paper published by Mok et al. for more information around upgrading your sepsis resuscitation by tailoring to micro and microcirculatory endpoints [7].
2. Obtain Microbial Cultures
The 2021 recommendation remains that cultures should be collected before antimicrobial administration as long as this will not result in any substantial delay. There are two noteworthy points around this recommendation.
The first is that the term ‘microbial’ culture serves as a nice reminder that we must consider multiple sources of infection beyond just the blood of our patients. This is particularly for our chronically unwell patients with indwelling lines or percutaneous drains.
The second point is that this iteration of the guideline defined ‘substantial delay’ as >45 minutes. While the evidence for this is based on expert opinion, it does provide us with a useful target until higher quality evidence can further refine this time point.
3. Administer Broad Spectrum Antimicrobials
A major driving point for the Surviving Sepsis Campaign’s strong recommendations for early antibiotics is the idea that mortality in sepsis is tied to time-to-antimicrobials.
While this is biologically plausible, critics of the Surviving Sepsis Campaign suggest that the recommendations are too strong for evidence derived from observational studies. More practically speaking, you might find yourself wondering just how dangerous is a delay in antibiotics?
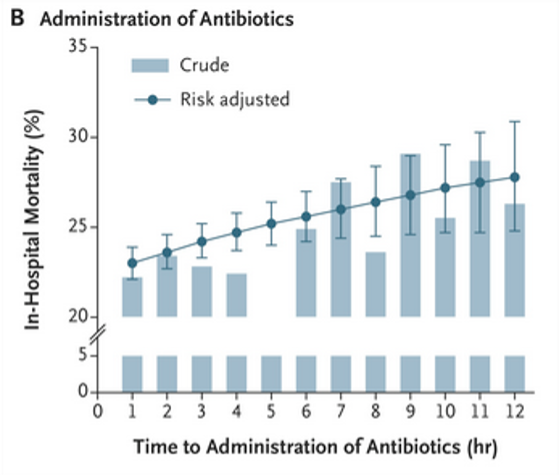
A compelling argument for early antibiotics comes from a large observational study by Seymour and co in NYC, published in the new England journal. This study population was unwell with most participants having septic shock.
As seen in this figure below, a steady temporal relationship seemed to be present [8].
Another retrospective study from 2017 found mortality to be tied to delay to antibotics, with the impact of antimicrobial delay most pronounced in those with shock [9].

Finally the highest calibre evidence we have on the topic comes from supplemental data derived from the MEDUSA trial – a multicenter RCT. While this trial’s primary outcome had nothing to do with time to antibiotics and mortality, in their supplemental information after correcting for multiple imputations for all patients in their study, they found a statistically significant 2% increase in mortality per hour of delayed antimicrobial initiation, p < 0.001 [10].
4. Crystalloid Resuscitation
What kind of fluid do I use?
The 2021 campaign kicks off its discussion of initial resuscitation with a strong recommendation for crystalloids. Give yourself a pat on the back, you’re probably already doing this.
Which Crystalloid is best?
Subsequently however, new to the 2021 guidelines, is a (weak) recommendation for balanced crystalloids (AKA Ringer’s Lactate) over normal saline.
Several studies have evaluated the saline vs ringer’s debate. I’m going to discuss the SMART trial which provided a major contribution to this recommendation. I won’t be discussing the SPLIT or SALT trials, as these trials had minimal septic patients, and given that this is clearly a unique sub-population, their results are less applicable. I will also discuss evidence which has been published since the guideline release – the BaSICS and PLUS trials.
SMART Trial
The SMART trial was a North American single centre RCT with multiple crossovers, enrolling approximately 15000 patients. They looked at a composite outcome of death, new dialysis or persistent renal dysfunction [11].
The primary outcome from the SMART trial favoured balanced crystalloids as you can see on the bottom row here. While the overall outcome favoured balanced crystalloids, the result was borderline. This, in addition to a composite outcome being used begs the question of clinical relevance, and many reasonably argue that this data alone does not settle the saline vs ringer’s debate [11].
More important for our discussion however, is the septic subgroup of patients. Here, the results more strongly favoured balanced crystalloids. Taking this a step further, a pre-planned secondary analysis of the septic sub-group of patients in the SMART trial was published in 2019.
They found a 4.9% absolute decrease in mortality in the balanced arm which was statistically significant (P = 0.01). Additionally, patients in the balanced group had better renal function leaving ICU, lower rates of dialysis, and were able to stop both vasopressors and dialysis earlier [12]. This data is in agreement with the findings of prior retrospective studies looking at the question of fluid choice in septic patients.
BaSICS Trial
In 2021 the BaSICS trial was published. This double-blinded RCT was carried out across 75 ICUs in Brazil, with a primary outcome of 90 day mortality and compared plasma-lyte with normal saline. A total of 10520 patients were enrolled and analyzed, with ~20% of these patient’s being septic.
This was a negative study, with no difference in mortality at 90 days.
A pre-specified sub-group analysis was planned for septic patient’s, which did not find any statistically significant mortality benefit. One could posit however, that the trend towards benefit seen in the sub-group analysis of the SMART trial persists, and that BaSICS simply wasn’t powered to identify it (HR 0.93; 95% CI 0.82 – 1.06) [13].
PLUS Trial
Most recently, in March 2022 the PLUS trial was published. This double-blind RCT compared Plasma-Lyte 148 (a balanced solution) to normal saline and evaluated 90 day mortality. They recruited 5037 patients from 53 ICUs across New Zealand and Australia.
This was again a negative study, finding no difference in rates of death, AKI or dialysis in patients receiving plasma-lyte.
Notably, ~40% of the patient’s in this study were septic. They found no difference in this subgroup of patient’s with respect to mortality and, unlike previous trials, there wasn’t an inkling of a trend towards benefit with balanced solution [14].
While initially hopeful that the Ringer’s vs Saline debate was coming to an end, the negative findings of BaSICS and PLUS lead me to be more skeptical of the sub-group data from the SMART trial.
The best evidence we have will likely arrive soon in the form of an individual patient data meta-analysis focused on the septic patient population. While it is impossible to know what the findings will be, my suspicion is that there will be a small (much smaller than seen in the SMART trial) but real benefit for patients with sepsis treated with balanced crystalloids.
How much fluid do I give?
For sepsis-induced hypoperfusion or septic shock, at least 30mL/kg of IV crystalloid is recommended in the first 3 hours of resuscitation. This obviously requires some clinical judgement based on the patient in front of you.
How many trials do you think there have been looking at the optimal fluid resuscitation strategy in sepsis?
Click here for answer / discussion…
The answer is 0. There has yet to be a prospective trial looking at different initial fluid resuscitation strategies in sepsis.
When we look to where the initial 30cc/kg came from and what evidence fostered this recommendation, it’s actually quite dogmatic.
The River’s trial first recommended 20-30cc/kg based on observational data that patient’s that received this amount of fluid did better[15].
When evaluating the translatability of the River’s trial, the PROCESS, PROMISE and ARISE studies used 30cc/kg [16–18]. On the basis of these four formative trials which guided the 21st century approach to sepsis care, 30cc/kg made its way into guidelines.
The situation becomes even more confusing when we look for guidance around the septic patient without hypoperfusion – there is no recommendation.
So what should we do? My recommendation would be to empirically provide 1-2L of fluid based on your initial assessment and re-evaluate your patient. I want to emphasize the re-evaluation.
While we don’t have evidence for benefit of a particular volume strategy, we do have evidence that resuscitation guided by lactate clearance, improving capillary refill and other dynamic parameters can improve mortality in sepsis patients.
Fortunately, further clarity should be provided to us in the near future on optimal fluid resuscitation strategy from the CLOVERS trial which is currently underway and expected to be published in early 2023.
5. Initiate Vasopressors
Which Agent do I start with?
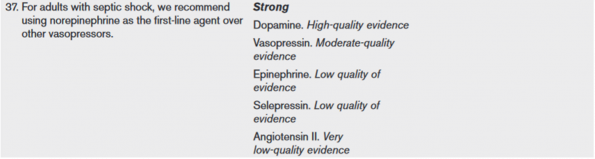
Use norepinephrine as your first line agent in sepsis. This is not a change, it’s what we’re already doing.
The surviving sepsis campaign provides a strong recommendation for this. I like that the 2021 campaign recognizes that while norepinephrine is clearly superior to dopamine, the evidence for superiority over other agents is much weaker [6].
Of note, there is now a recommendation to initiate vasopressors peripherally in patients with septic shock, rather than waiting for central venous access. This is great from the perspective of guidelines supporting actual practice, as this is something we do frequently in the ED.
What Map do I target?
In the absence of high quality evidence, the surviving sepsis campaign in 2012 suggested we use a target MAP of 65 based on expert consensus. The answer to this question has since been informed by two major RCTs.
The first trial by Asfar et al. was a multicenter open label trail evaluating a high pressure group (MAP 80-85) vs a low pressure group (MAP 65-70). Their primary outcome was 28 day mortality. They found no difference in mortality between strategies. They did however note a statistically significant 4% increased incidence of atrial fibrillation in the high target group, presumably due to increased catecholamine exposure [19]. Despite the findings of the paper by Asfar et al., many clinicians were concerned that a MAP of 65 might prove harmful in geriatric patients with chronic hypertension.
This prompted the 65 trial, published in 2020. This study sought to evaluate the impact of a MAP target of 65 on our elderly hypertensive patients. This was a well done, prospective, multi-center RCT. The majority of the patients in this trial were ICU patients with sepsis or septic shock. A MAP of 65 was compared with usual care – which worked out to a MAP of ~75.
They found no statistically significant difference in mortality between arms, with a trend to better survival in those with a MAP target of 65. Furthermore, they found no difference in the incidence of renal failure, arrhythmia or other severe adverse events [20].
Thus, based on these trials, we should continue to use a MAP target of 65 for our septic patients. Additionally, we should feel comfortable doing so, even in geriatric patients with histories of chronic hypertension.
Which agent do we initiate if we find our patient unresponsive to norepinephrine?
The 2021 surviving sepsis guideline authors performed their own systematic review to answer this question. They reviewed 10 RCTS on the subject. They looked at rates of death dialysis and dysrhythmia with the addition of vasopressin to norepi.
They report a statistically significant benefit for mortality in vasopressin + norepi and no effect on dialysis or dysrhythmia rates with vasopressin use [6].
I however have multiple issues with the results of their systematic review:
- They were not published independently.
- There is no citation provided on the numbers they provide.
- There is no mention of it on prospero, and they do not detail their methods.
- The 10 studies used are mentioned in their supplemental information. I’ve reviewed these 10 studies individually and only 2 of them look directly at this question where one of the trials was looking at post op, non septic patients.
This is problematic, as there are not any meta-analyses that otherwise directly answer this question. Given this critical limitation, we must be skeptical of this data.
Looking instead at the primary literature on vasopressin and norepinephrine, there are only two RCTs that look to answer the question of if adding vasopressin as a second line agent is beneficial for our patients. One of these studies was an unregistered RCT presented at a critical care conference in Brazil and never published.
The other, is the VAAST trial published in the New England Journal in 2008. This was a Canadian and Australian multi-center RCT. The study looked at patient’s already receiving 5mcg/min of norepinephrine, and compared the impact of further norepinephrine to the addition of vasopressin [21].
In this trial, there was absolutely no difference in adverse event rates between arms. Additionally, while both 28 and 90 day mortality figures favoured vasopressin + norepinephrine over norepinephrine alone, neither were statistically significant.
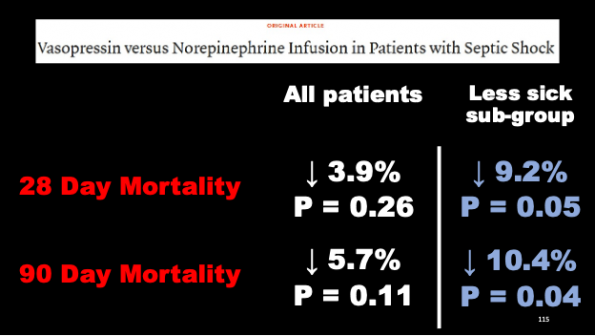
However, when we look at the subgroup of patients with lower norepinephrine requirements at randomization (5-14mcg/min), a signal of benefit for mortality becomes apparent. This signal is made more interesting by the fact that the study was powered only to detect a 10% mortality difference [21].
So that brings us back to the question of what do we use as our second line agent? The 2021 surviving sepsis campaign recommends vasopressin.
I think the evidence for this is exceptionally weak; the subgroup analysis of one trial is the only data for a significant benefit for vasopressin over increasing doses of norepinephrine alone.
Conversely, there is biologic plausibility to using an agent that affects a different receptor, we at least have data to suggest there is not an increased risk of harm and if we are to suspend skepticism of the campaign author’s systematic review, there may be a small mortality benefit.
On this basis, I would recommend considering vasopressin as a second line agent.
If you’ve decided you’re going to use vaso as a second line agent, at what point do you pull that trigger and start it?
There is no high quality evidence to guide this decision. Based on expert consensus the 2021 surviving sepsis campaign recommends doing this when norepinephrine doses are in the range of 15-30mcg/min.
Given that the best evidence we actually have on this topic is from the VAAST trial, which we just reviewed, and that the group which actually showed a benefit were those with lower norepinephrine requirements, I would suggest initiating it at lower norepinephrine concentrations.
My recommendation would thus be that if you are going to initiate vasopressin in a patient with septic shock, that this is done once norepinephrine infusion reaches 10-15mcg/min
___
I look forward to hearing your thoughts on my summary and addressing any questions or criticisms you might have. Leave a comment below or tweet me directly
[1] Singer M, Deutschman CS, Seymour C, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). JAMA – J Am Med Assoc 2016;315:801–10. https://doi.org/10.1001/jama.2016.0287. [2] Fernando SM, Tran A, Taljaard M, Cheng W, Rochwerg B, Seely AJE, et al. Prognostic accuracy of the quick sequential organ failure assessment for mortality in patients with suspected infection, A systematic review and meta-Analysis. Ann Intern Med 2018;168:266–75. https://doi.org/10.7326/M17-2820. [3] Serafim R, Gomes JA, Salluh J, Póvoa P. A Comparison of the Quick-SOFA and Systemic Inflammatory Response Syndrome Criteria for the Diagnosis of Sepsis and Prediction of Mortality: A Systematic Review and Meta-Analysis. Chest 2018;153:646–55. https://doi.org/10.1016/j.chest.2017.12.015. [4] Herwanto V, Shetty A, Nalos M, Chakraborty M, McLean A, Eslick GD, et al. Accuracy of Quick Sequential Organ Failure Assessment Score to Predict Sepsis Mortality in 121 Studies Including 1,716,017 Individuals. Crit Care Explor 2019;1:e0043. https://doi.org/10.1097/cce.0000000000000043. [5] Levy MM, Evans LE, Rhodes A. The Surviving Sepsis Campaign Bundle. Crit Care Med 2018;46:997–1000. https://doi.org/10.1097/CCM.0000000000003119. [6] Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Crit Care Med 2021;49:e1063–143. https://doi.org/10.1097/CCM.0000000000005337. [7] Mok G, Hendin A, Reardon P, Hickey M, Gray S, Yadav K. Macrocirculatory and Microcirculatory Endpoints in Sepsis Resuscitation. J Intensive Care Med 2021;36:1385–91. https://doi.org/10.1177/0885066620982585. [8] Seymour CW, Gesten F, Prescott HC, Friedrich ME, Iwashyna TJ, Phillips GS, et al. Time to Treatment and Mortality during Mandated Emergency Care for Sepsis. N Engl J Med 2017;376:2235–44. https://doi.org/10.1056/nejmoa1703058. [9] Liu VX, Fielding-Singh V, Greene JD, Baker JM, Iwashyna TJ, Bhattacharya J, et al. The timing of early antibiotics and hospital mortality in sepsis. Am J Respir Crit Care Med 2017;196:856–63. https://doi.org/10.1164/rccm.201609-1848OC. [10] Bloos F, Rüddel H, Thomas-Rüddel D, Schwarzkopf D, Pausch C, Harbarth S, et al. Effect of a multifaceted educational intervention for anti-infectious measures on sepsis mortality: a cluster randomized trial. Intensive Care Med 2017;43:1602–12. https://doi.org/10.1007/s00134-017-4782-4. [11] Semler MW, Self WH, Wanderer JP, Ehrenfeld JM, Wang L, Byrne DW, et al. Balanced Crystalloids versus Saline in Critically Ill Adults. N Engl J Med 2018;378:829–39. https://doi.org/10.1056/nejmoa1711584. [12] Brown RM, Wang L, Coston TD, Krishnan NI, Casey JD, Wanderer JP, et al. Balanced Crystalloids versus Saline in Sepsis: A secondary analysis of the SMART clinical trial. Am J Respir Crit Care Med 2019;200:1487–95. https://doi.org/10.1164/rccm.201903-0557OC. [13] Zampieri FG, Machado FR, Biondi RS, Freitas FGR, Veiga VC, Figueiredo RC, et al. Effect of Intravenous Fluid Treatment with a Balanced Solution vs 0.9% Saline Solution on Mortality in Critically Ill Patients: The BaSICS Randomized Clinical Trial. JAMA – J. Am. Med. Assoc., vol. 326, American Medical Association; 2021, p. 818–29. https://doi.org/10.1001/jama.2021.11684. [14] Finfer S, Micallef S, Hammond N, Navarra L, Bellomo R, Billot L, et al. Balanced Multielectrolyte Solution versus Saline in Critically Ill Adults. N Engl J Med 2022;386:815–26. https://doi.org/10.1056/nejmoa2114464. [15] Rivers E, Nguyen B, Havstad S, Ressler J, Muzzin A, Knoblich B, et al. Early Goal-Directed Therapy in the Treatment of Severe Sepsis and Septic Shock. N Engl J Med 2001;345:1368–77. https://doi.org/10.1056/nejmoa010307. [16] Mouncey PR, Osborn TM, Power GS, Harrison DA, Sadique MZ, Grieve RD, et al. Trial of Early, Goal-Directed Resuscitation for Septic Shock. N Engl J Med 2015;372:1301–11. https://doi.org/10.1056/nejmoa1500896. [17] Goal-Directed Resuscitation for Patients with Early Septic Shock. N Engl J Med 2014;371:1496–506. https://doi.org/10.1056/nejmoa1404380. [18] A Randomized Trial of Protocol-Based Care for Early Septic Shock. N Engl J Med 2014;370:1683–93. https://doi.org/10.1056/nejmoa1401602. [19] Asfar P, Meziani F, Hamel J-F, Grelon F, Megarbane B, Anguel N, et al. High versus Low Blood-Pressure Target in Patients with Septic Shock. N Engl J Med 2014;370:1583–93. https://doi.org/10.1056/nejmoa1312173. [20] Lamontagne F, Richards-Belle A, Thomas K, Harrison DA, Sadique MZ, Grieve RD, et al. Effect of Reduced Exposure to Vasopressors on 90-Day Mortality in Older Critically Ill Patients with Vasodilatory Hypotension: A Randomized Clinical Trial. JAMA – J Am Med Assoc 2020;323:938–49. https://doi.org/10.1001/jama.2020.0930. [21] Russell JA, Walley KR, Singer J, Gordon AC, Hébert PC, Cooper DJ, et al. Vasopressin versus Norepinephrine Infusion in Patients with Septic Shock. N Engl J Med 2008;358:877–87. https://doi.org/10.1056/nejmoa067373.References